
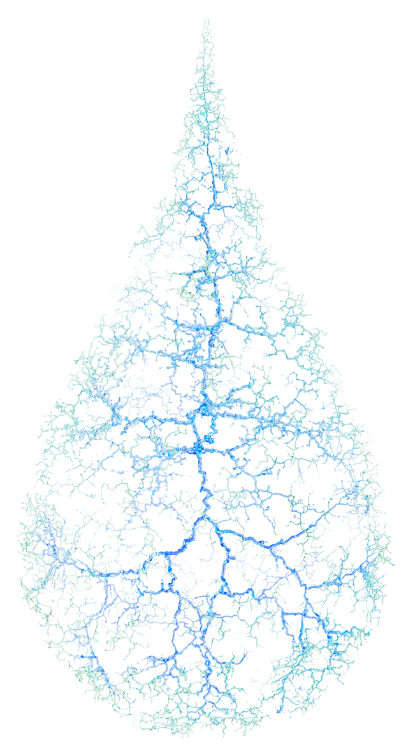
A UNIQUE MOA TARGETS CORNEAL NERVE DAMAGE, THE UNDERLYING CAUSE OF NK1-4
Mechanism of Action
Cenegermin-bkbj, the active ingredient in OXERVATE®, is a recombinant form of human nerve growth factor (rhNGF)1
NGF is an endogenous protein involved in the differentiation and maintenance of neurons, which acts through specific high-affinity (ie, TrkA) and low-affinity (ie, p75NTR) nerve growth factor receptors in the anterior segment of the eye to support corneal innervation and integrity1
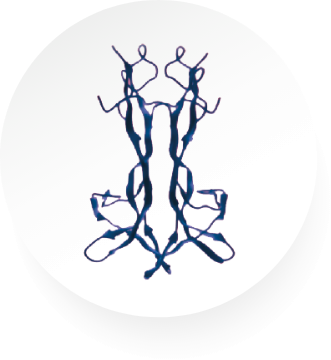
Cenegermin-bkbj
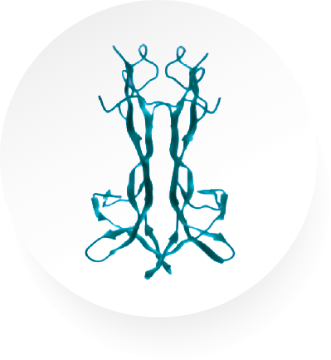
Endogenous NGF
Cenegermin-bkbj is structurally identical to human NGF protein made in ocular tissues.5
Download an overview of neurotrophic keratitis (NK) and OXERVATE®



How NGF Works
NGF and the ocular surface
Endogenous NGF supports corneal integrity through 3 mechanisms contributing to ocular surface homeostasis (shown in preclinical models)1,2,6:
- Corneal innervation
- Tear secretion
- Epithelial cell growth
As part of ocular surface homeostasis, corneal epithelial cells produce NGF to support sensory nerve health, and sensory nerves produce neuromediators to support corneal epithelial cell health.2

