What is OXERVATE®?
The only prescription eye drop FDA‑approved to treat neurotrophic keratitis.
The only prescription eye drop FDA‑approved to treat neurotrophic keratitis.


Cenegermin-bkbj is a manufactured form of a natural protein in the body called nerve growth factor (NGF).
In 2 clinical trials, 151 patients with neurotrophic keratitis (NK) were treated with OXERVATE or placebo (inactive substance) for 8 weeks.
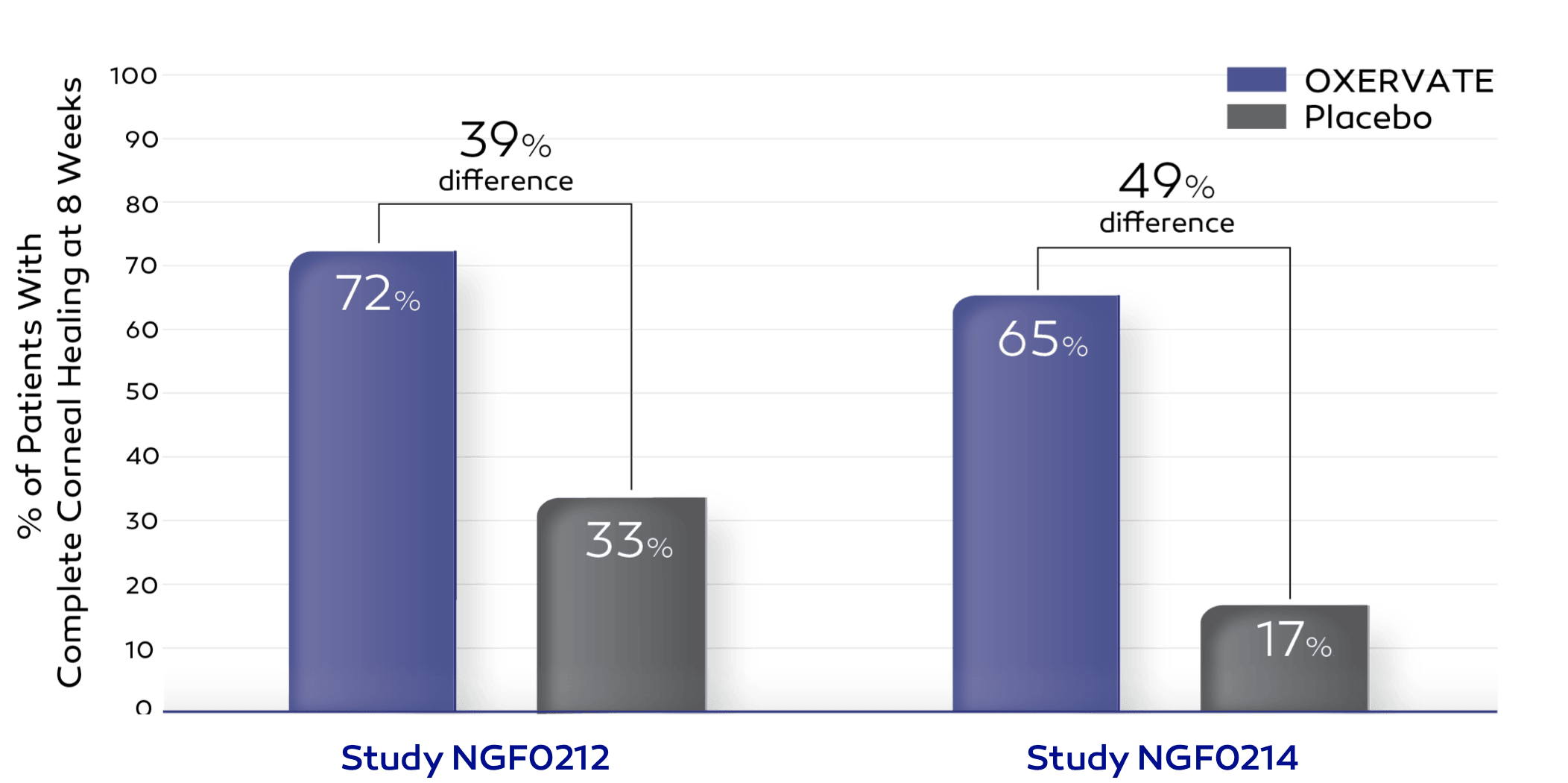
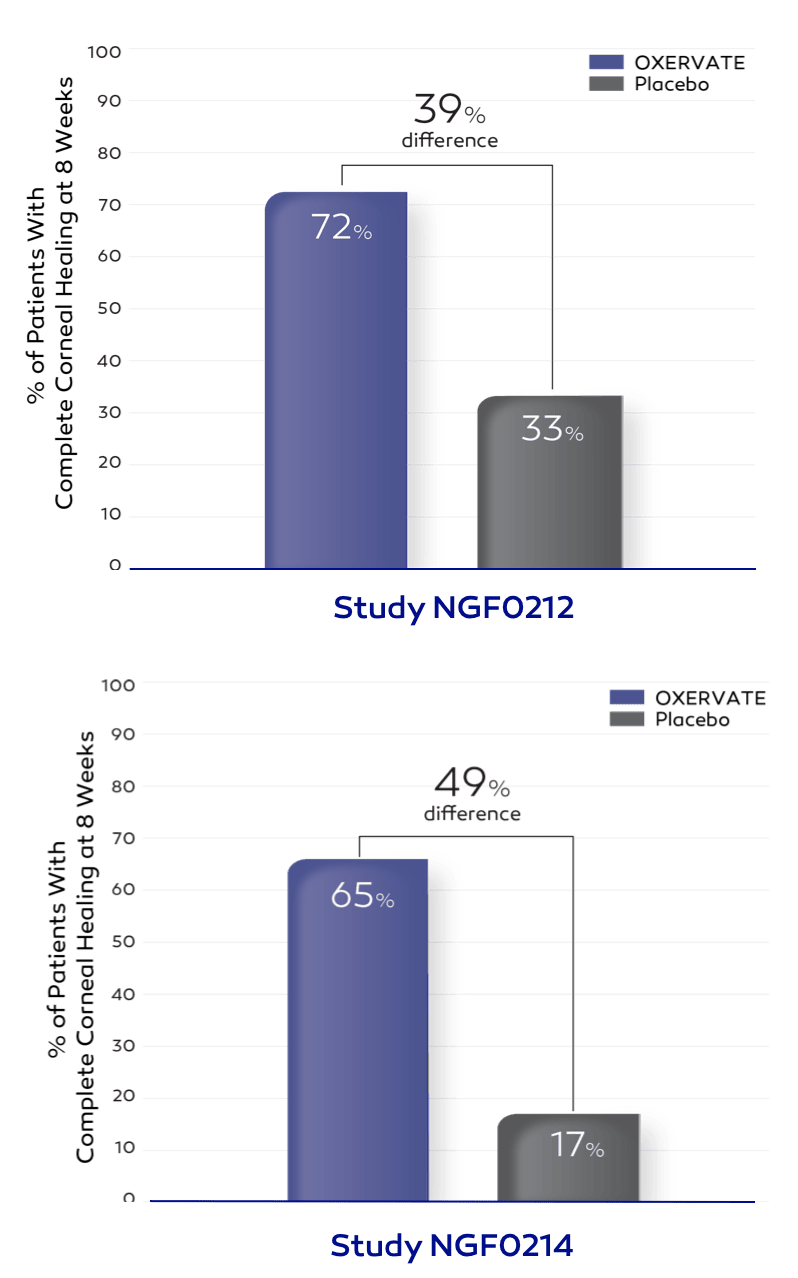
*At 8 weeks, each patient’s degree of corneal healing was determined by a test to assess damage to the cornea (corneal staining). If there was no staining in the damaged part of the cornea, and no persistent staining in the rest of the cornea, the cornea was considered completely healed (complete corneal healing).
More patients who received OXERVATE experienced complete corneal healing compared to those who received placebo (72% vs 33% and 65% vs 17%).
The most common side effect with OXERVATE was eye pain (~16%). Other side effects with OXERVATE, in 1% to 10% of patients, included corneal deposits, feeling of having something in the eye, enlarged blood vessels in the white of the eyes, swelling of the eye, sensitivity to light, increase of tears, and headache.
What are the possible side effects of OXERVATE?
This image is for illustrative purposes only. This is not an exact representation of a patient.
Call your doctor for medical advice about side effects.
You may report side effects to FDA at 1-800-FDA-1088
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use OXERVATE for a condition for which it was not prescribed. Do not give OXERVATE to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or doctor for information about OXERVATE that is written for health professionals.
For more information about OXERVATE talk to your doctor or pharmacist.

You may have questions when you’re prescribed OXERVATE.
Download the OXERVATE Brochure and use the Doctor Discussion Guide within during your next appointment.
Sign up to receive more information, including tools and resources for accessing and using OXERVATE.
ALL FIELDS REQUIRED
Before you use OXERVATE, tell your doctor about all of your medical conditions, including if you:
How should I use OXERVATE?
What should I avoid while using OXERVATE?
What are the possible side effects of OXERVATE?
Tell your doctor if you have any side effects that bother you. These are not all the possible side effects of OXERVATE.
For more information about OXERVATE talk to your healthcare provider or pharmacist.
To report ADVERSE REACTIONS, contact Dompé U.S. Inc. at 1-833-366-7387 or FDA at
1-800-FDA-1088 or www.fda.gov/medwatch.
What is OXERVATE?
Please see full Prescribing Information and Patient Information for OXERVATE.